Deal: Biogen to co-develop and co-commercialize LRRK2 inhibitors with Denali Therapeutics
First-in-class drugs for Parkinson’s disease
The “Deal” series of articles discuss business agreements and the implications. These articles offer unique and integrated perspectives to the significance of the agreements.
Two deals with two biotechnology giants
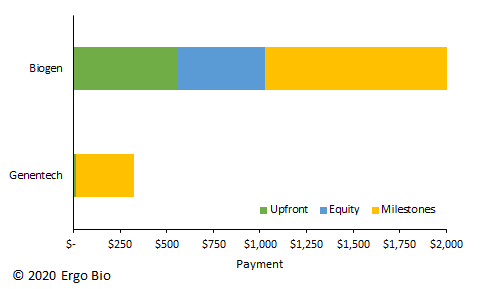
Early last month, Biogen ($BIIB) announced an agreement with Denali Therapeutics ($DNLI) to co-develop and co-commercialize small-molecule inhibitors of leucine-rich kinase 2 (LRRK2) for Parkinson’s disease (ref). Biogen also reserved exclusive option rights to the antibody-based, blood-brain barrier transport technology from Denali. The terms of the agreement included an upfront payment of $560M, an equity investment of $465M, and milestone payments up to $1.125B. This deal will help Denali advance its LRRK2 program further into clinical trials and continue expanding its therapeutic pipeline around neurodegeneration.
To understand the significance of this deal, it is necessary to review the origins of the LRRK2 program. In 2015, numerous directors and scientists left Genentech to create Denali and develop solutions for neurodegeneration (ref). Denali was launched with $217M from investors like ARCH Venture Partners, Fidelity Biosciences Research Initiative, and Flagship Ventures (ref). The Denali co-founders included Marc Tessier-Lavigne, Ryan Watts, Alex Schuth, all of whom had considerable responsibilities at Genentech. With an all-star team, Denali quickly IPO’d in 2017 with a valuation of $1.7B (ref). The S-1 filing revealed an exclusive license agreement with Genentech’s preclinical LRRK2 program (ref). In 2016, Denali paid an upfront fee of $8.5M and technology transfer fee of $1.5M. This $10M agreement also included milestone payments up to $315M and royalties on net sales. In considering the recent Biogen agreement, Denali effectively traded a call option on the LRRK2 program with Genentech and later on secured a massively successful deal. Why was it that Genentech so readily gave up the LRRK2 program? And why does Biogen want to pick up the LRRK2 program?
Challenges for LRRK2 inhibitors
Historically, there has been limited success with developing effective treatments for patients with neurodegeneration, including Parkinson’s disease. Parkinson’s disease is the most prevalent movement disorder and is characterized by tremors and bradykinesia. Recent discoveries of genetic variations associated with Parkinson’s disease have instigated the development of novel drugs (ref). These include α-synuclein, glucocerebrosidase (GBA), and LRRK2. Despite having better understanding of the genetic risks for Parkinson’s disease, there is a dearth of clinical data, including proof of concept, on whether targeted therapies could be efficacious.
Source: Roosen and Cookson 2016
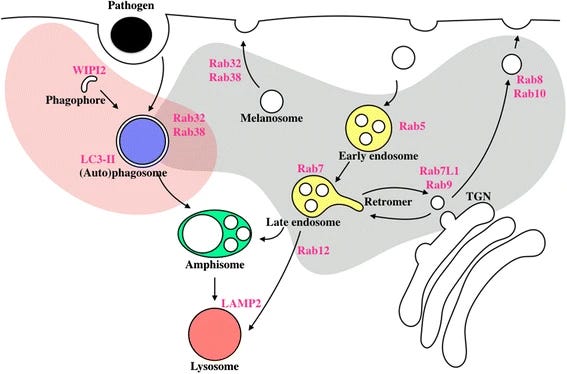
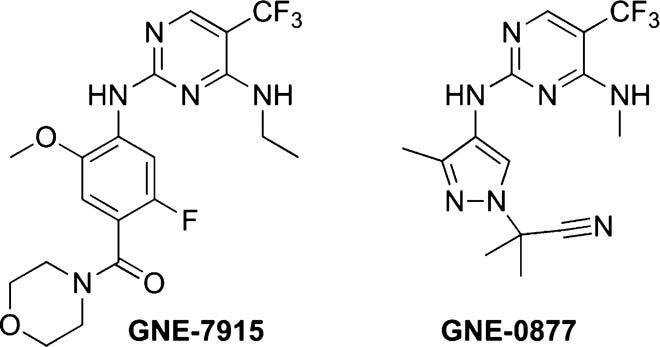
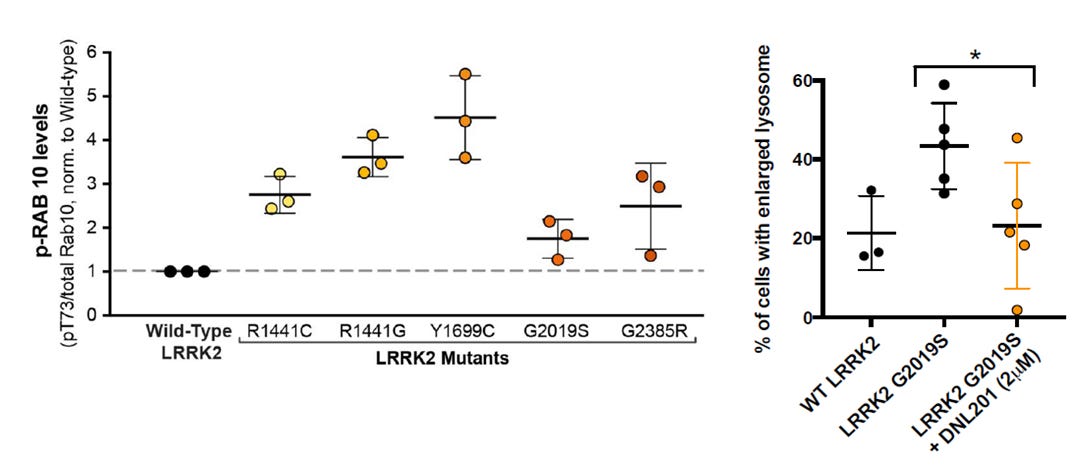
LRRK2 has a complex role in the regulation of lysosomal storage, endocytosis, and autophagy (ref). The aberrant signaling of LRRK2, either by overexpression or genetic variation, is strongly implicated in the pathogenesis of familial and idiopathic Parkinson’s disease (ref). Its mutations are pervasive among patients with Parkinson’s disease, and one such variant (G2019S) has been described to be a cause of the idiopathic form via Mendelian inheritance (ref). Hence, LRRK2 is a promising therapeutic target. Yet, small-molecule inhibitors of LRRK2 have only recently entered clinical trials. In 2012, Genentech published work on the discovery of potent and brain-penetrant LRRK2 inhibitors, including GNE-7915 (ref). However, these aminopyrazole molecules exhibited poor solubility and could give rise to undesirable metabolites. Genentech later improved on the safety profile by replacing the aniline functionality on the aminopyrazole molecules (ref).
The safety of LRRK2 inhibitors is concerning due to preclinical evidence of anomalous tissue phenotypes. Rodents with LRRK2 knock-out present phenotypic changes in pulmonary and renal tissue, specifically an accumulation of lysosomes in kidney proximal tubule epithelial cells and lamellar bodies in lung type II cells (ref). Rodents expressing LRRK2-dead variant had similar abnormalities in renal tissue but not pulmonary tissue. Interestingly, there is slight hypertrophy of the kidney in male, but not female, rodents with LRRK2 knock-out. Importantly, there was not significant neuropathology in the knock-out rodents. This murine study hints at the potential of LRRK2-associated toxicity in multiple tissues, and that histopathological differences could arise depending on how LRRK2 signaling is suppressed. The phenotypic findings in rodents with LRRK2 knock-out were independently reproduced by Genentech.
Source: Fuji et al. 2015
Genentech studied the activity of their LRRK2 inhibitors, GNE-7915 and GNE-0877, in animals (ref). These molecules had low nanomolar Kis and IC50s, and exhibited high specificity determined by an off-target panel of other kinases and receptors. Rodents administered with these inhibitors did not result in pulmonary or renal pathology. Notably, both inhibitors were present in the lungs and kidneys at total concentrations close to that of the brain. GNE-7915 had a slightly lower unbound brain/unbound plasma ratio than that of GNE-0877. Regardless, phosphorylation profiling indicated that both molecules mediated LRRK2 inhibition in the brain, lungs, and kidneys despite differences in exposure. Interestingly, GNE-0877 was not well tolerated in a 7-day murine trial, whereas GNE-7915 was well tolerated (up to 20-fold higher than the IC50). Genentech proceeded to evaluate repeat dosing of GNE-7915 in cynomolgus monkeys over 7 days. The monkeys maintained their body weight, but exhibited tremors and hypoactivity at high doses. These clinical symptoms were transient and resolved by the subsequent dose. Importantly, GNE-7915 appeared to enhance vacuolation and the number of lamellar bodies in pulmonary tissue. This was in both sexes and when administered with >25 mg/kg per day. The brain exposure of GNE-7915 was very high and there was evidence of reduced LRRK2 phosphorylation in brain tissue at all doses. Genentech then compared the toxicity of GNE-7915 and GNE-0877 in monkeys across 29 days. Interestingly, no clinical pathology was observed with GNE-0877, while phenotypic changes in the lungs were again observed with GNE-7915. No effect on renal function was observed. While renal toxicity was not of concern, pulmonary toxicity is likely the primary issue for the safety profile of LRRK2 inhibitors.
In the same year that this study was published, multiple directors and employees left Genentech to form Denali. The neuroscience department was split into factions and Denali was effectively seceding from Genentech. It was clear that Genentech was not interested in continuing the LRRK2 program, but key individuals involved with the studies had serious convictions. Despite the contentions that surrounded the LRRK2 program, this is not too surprising. Neurodegenerative diseases are incredibly challenging to treat and drug development is immensely expensive. Evidently, this makes big pharmaceutical companies wary of spending valuable resources on high-risk programs. As an unrelated example, Pfizer recently closed their neuroscience division and halted their programs in Alzheimer’s and Parkinson’s diseases (ref). Despite the challenges, Denali was founded to discover solutions for patients with neurodegenerative diseases and change their lives. With these massive, unmet clinical needs, successful drug development will also be quite lucrative.
First-in-human trials
Source: Denali’s R&D Day in 2018
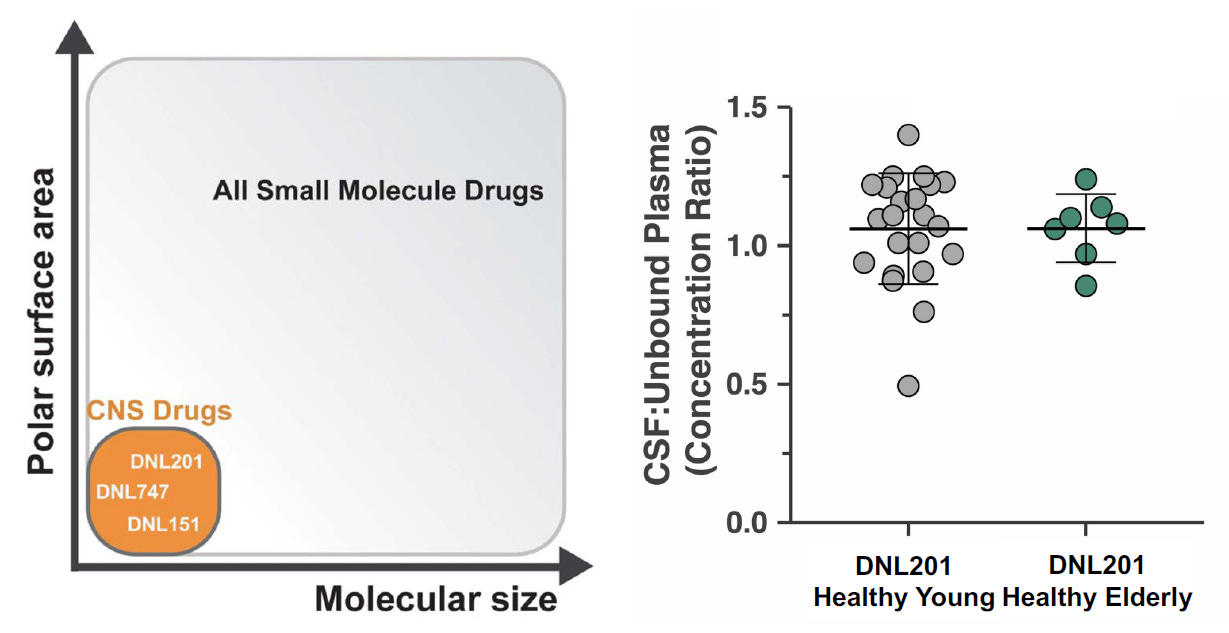
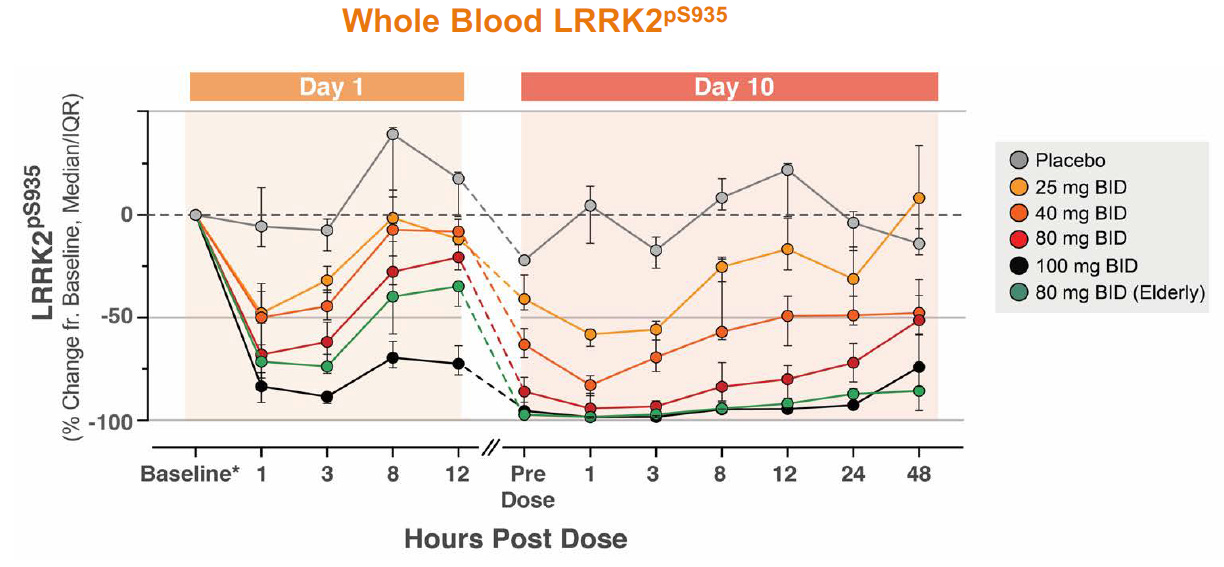
Denali has multiple drug candidates for LRRK2 and decided to investigate two molecules (DNL201 and DNL151) in early clinical trials. Due to preclinical evidence of toxicity, the FDA had placed an exposure cap until review of early safety data from the phase 1 trials. This partial clinical hold was soon lifted as DNL201 demonstrated considerable suppression of LRRK2 without significant toxicity (ref). In healthy volunteers, DNL201 offered >90% LRRK2 inhibition at peak levels and >50% LRRK2 inhibition at trough levels. LRRK2 inhibition was evaluated by blood biomarker assays of LRRK2 phosphorylation at S935 and Rab10 phosphorylation. Note, Rab10 is a substrate of LRRK2 and both are highly expressed in neutrophils and monocytes, so this enables robust evaluation of drug activity from blood measurements (ref). As a separate note, but importantly, DNL201 appeared to have considerable brain exposure in both young and elderly volunteers.
Source: Denali’s R&D Day in 2018
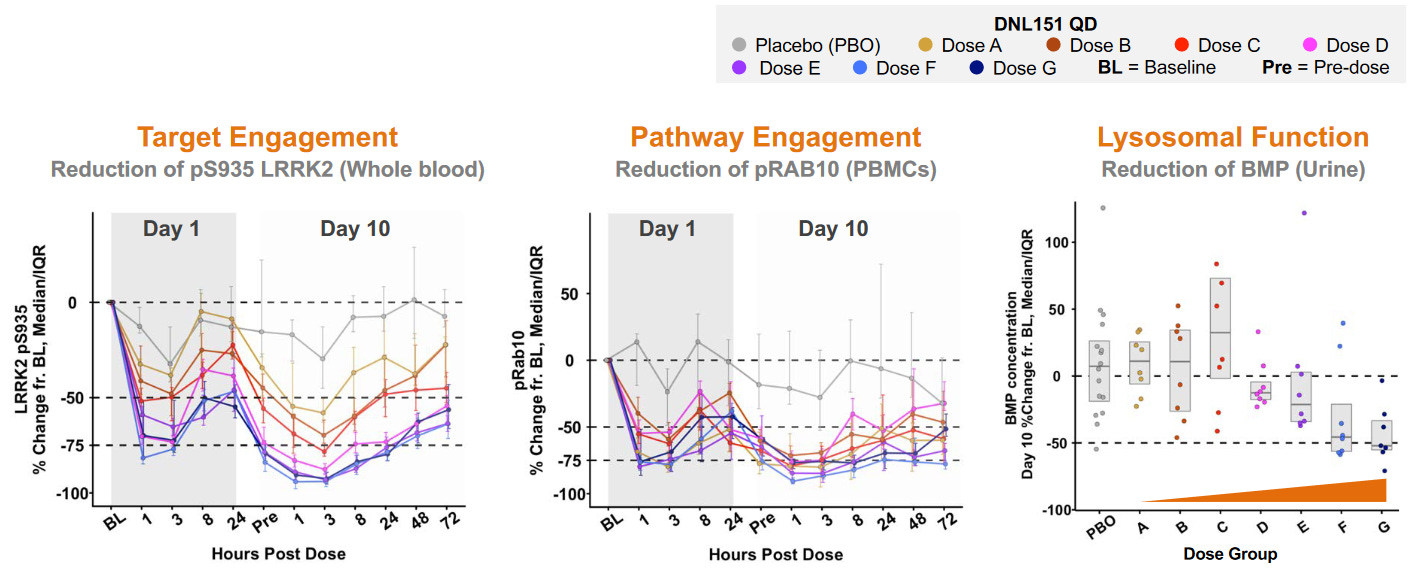
While also conducting a phase 1 study of DNL151 in the Netherlands, Denali had completed the initial dose escalation study of DNL201 with >100 healthy volunteers (ref). Denali then arranged a collaboration with CENTOGENE for identifying and recruiting patients with LRRK2 mutations (ref). LRRK2 variations are known to have distinctly different activity, thus plans for patient stratification can be developed as clinical data emerges. Denali continues to build on their network for patient enrollment, including a website to keep up to date on their ongoing clinical trials (engageparkinsons.com).
Source: Denali’s R&D Day in 2018
Early this year, Denali announced positive results from the phase 1b study of DNL201 and that all safety objectives were met in the phase 1 study of DNL151 (ref). Moreover, it appears that biomarker goals were also met and that DNL151 has dose-dependent activity in healthy volunteers. In August, Denali chose to advance DNL151 into late-stage clinical trials due to its pharmacokinetic attributes (ref). On the same day, Denali announced the deal with Biogen (ref). The plan is to conduct two studies in parallel to evaluate Parkinson’s disease patients with and without LRRK2 mutations.
Source: Denali’s corporate presentation at the Morgan Stanley conference
Biogen paid a steep price to be involved with the co-development and co-commercialization of the LRRK2 program, and quite early in its development. They may have been convinced by clinical data that hasn’t yet been released to the public. It’s also likely to add another shot on goal to their arsenal of neurodegeneration solutions, such as aducanumab for Alzheimer’s disease (ref). My friend, Richard Murphey of Bay Bridge Bio, has analyzed Biogen’s decision to submit aducanumab for regulatory approval despite announced plans to cancel the program (ref). This is worth the read; he discusses the background on FDA submission, what to consider when evaluating clinical data, and the limitations of post-hoc analyses.
Will it work?
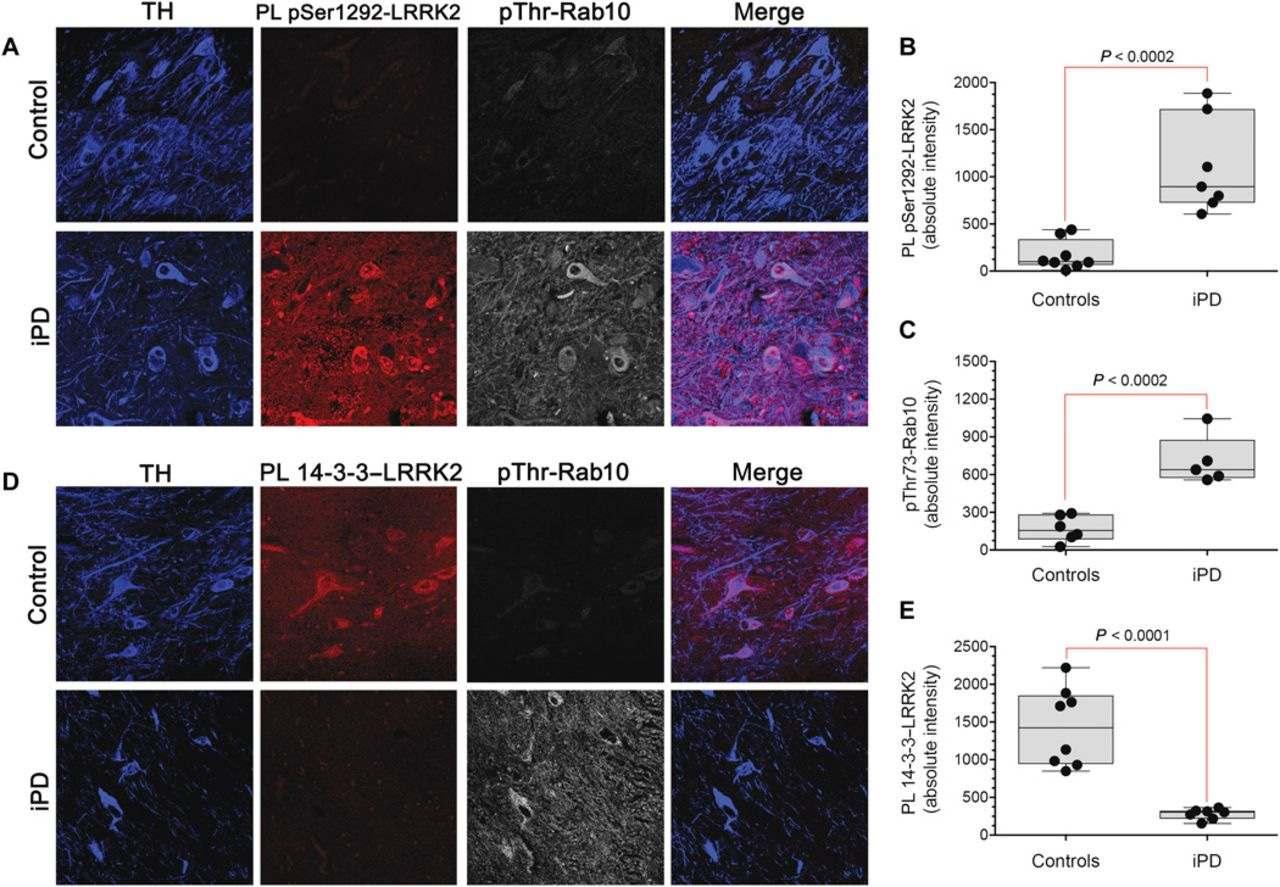
It’s difficult to have a strong opinion on whether DNL151 will be approved this early in clinical development. The initial clinical data of DNL151 and DNL201 in healthy volunteers are promising and demonstrate activity based on multiple biomarkers. The brain exposure of these molecules are quite high. However, blood measurements are not necessarily indicative of drug activity in the brain. That said, the scientific evidence for LRRK2 involvement in the pathogenesis of Parkinson’s disease is strong. LRRK2 is known to promote aggregation and autoubiquitination because of its interaction with parkin, an E3 ligase (ref). LRRK2 aberrations are known to cause neurite shortening and induce autophagic vacuoles (ref). Post-mortem studies of patients with idiopathic Parkinson’s disease have revealed enhanced levels of LRRK2 autophosphorylation and Rab10 phosphorylation (ref).
Source: Di Miao et al. 2018
From a management perspective, Denali is performing exceedingly well. The startup has explored two molecules in humans, with many more as backup drug candidates. Multiple biomarkers were identified to guide dosing and potentially patient stratification. Denali has also arranged an extensive network for patient enrollment and genotyping. Even with a large pipeline, Denali is well capitalized and the deal with Biogen derisks them financially. It will be very exciting to watch Denali as they make progress on the LRRK2 program. While they are certainly pioneers in the space, there are also rumors of other startups working on LRRK2 inhibitors.
Author information
Ergo Bio closely follows innovation in the biotechnology space and evaluates interesting drugs and deals. It is run by Vandon T Duong (LinkedIn), feel free to connect! I am a biotech enthusiast and a molecular engineer by training. I am also an avid consumer of news and research around precision medicine.
Ergo Bio pages
Disclaimer
This article serves informational purposes only and should be treated strictly as educational material, not as investment recommendation or legal advice. The information presented may be inaccurate or out-of-date. The contributing authors and editors disclaim liability for any errors or omissions. Any opinions expressed may change without notice. Ergo Bio LLC reserves all rights to the content generated through this resource hub (Ergo Bio Insights).