Advances: mRNA vaccines are on the verge of realization
COVID-19 caused a monumental paradigm shift in vaccinology
The “Advances” series of articles discuss technological breakthroughs in the life sciences. These articles present important findings from scientific studies and other resources.
Highlights and takeaways
A personal anecdote about the time I learned about Moderna and the promise of mRNA products directly from the CEO.
A brief dive into the technology behind mRNA-1273 and mRNA vaccines. Interestingly, the major mRNA vaccine companies are likely using lipid nanoparticle formulations with overlapping IP space.
Positive readouts from three vaccine candidates in phase 3 trials have been announced! A quick explanation of the vaccine efficacy results, back-of-the-envelope calculations, and the implications.
A personal anecdote
First of all, Happy Thanksgiving! I hope everyone had a nice holiday and was able to spend time with their loved ones. Remember to work off the Thanksgiving meals :)
Source: Moderna corporate presentation
I first heard about Moderna and mRNA vaccines when Stéphane Bancel visited the biomolecular engineering course taught by Ben Hackel. Bancel was a graduate from the chemical engineering program at the University of Minnesota and studied macroporous microcarriers with Wei-Shou Hu (ref). I was in another class taught by Hu and he mentioned how Bancel became quite an enigma in the biotechnology community. Earlier that year, STAT News posted an investigative report describing the intense, secretive work culture at Moderna (ref).
Source: STAT News report
I attended Bancel’s talk about the future of delivering synthetic mRNA as instructions to the protein factories within cells of the patient. The idea was quite compelling. Manufacturing mRNA is relatively faster and cheaper than directly synthesizing the proteins, and one could encapsulate the transcripts within lipid nanoparticles to mitigate degradation by extracellular RNases. (Anyone that has done bench science knows that RNase is everywhere!) Furthermore, the protein produced by the patient themselves would have proper post-translational modifications. Overall, Bancel had a strong vision for the future of mRNA products and was able to clearly convey it, even to an audience of university students. Despite his tight schedule, I was able to catch a few minutes with Bancel and (pardon the cliché) was further inspired to pursue my interest in biotechnology business.
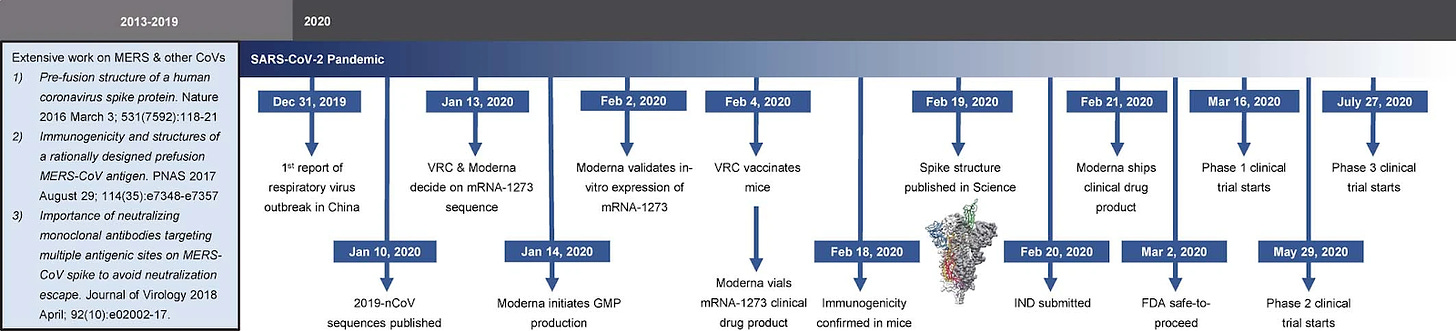
At the end of 2018, Moderna ($MRNA) raised over $600M during its IPO, with a valuation of approximately $7.5B (ref). Today, the market cap of Moderna exceeds $50B, rising over 600% year-to-date. As everyone knows, this swelling in valuation is due to their rapid vaccine development for COVID-19. What some may not be aware of is that this year also marks a stark change in the transparency of their business activities. Moderna released a full set of confidential protocols for the clinical study of mRNA-1273 (ref). The company also reorganized their scientific publications page with clear explanations of what these reports included (ref). Moderna has transformed from having ultra secret practices to a (somewhat) better understood biotechnology operation.
The technology behind mRNA-1273 and other mRNA vaccines
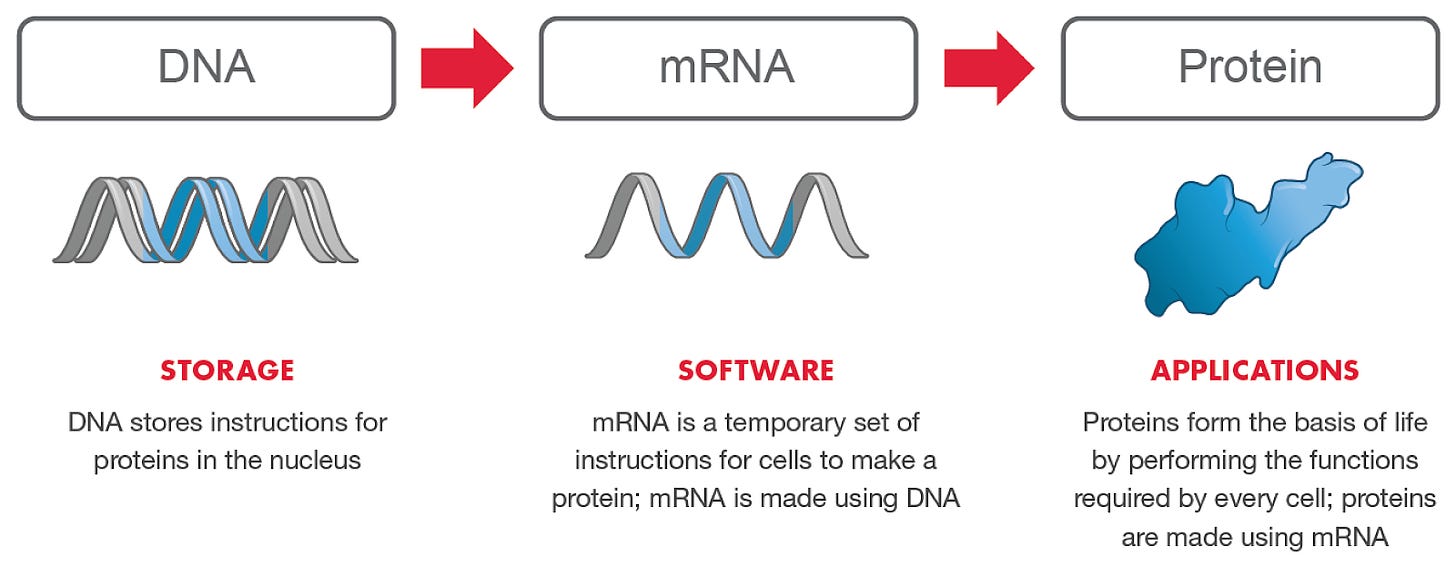
The purpose of vaccines is to elicit an immune response and facilitate acquired immunity against an infectious disease. Traditionally, vaccines were categorized as inactivated (previously virulent), attenuated (live but weakened), or subunit (protein fragment). mRNA vaccines have been under scientific study, but none have been clinically approved to date (ref). There are several advantages to mRNA vaccines, including its non-infective and non-integrating nature, tunable half-life and immunogenicity, and ease-of-manufacturing. Moreover, RNA is an activator of toll-like receptors (TLRs) and chemical modifications can alter the degree to which it stimulates the immune system (ref). As such, multiple biotech startups have been launched to develop mRNA products, including Moderna, BioNTech ($BNTX), CureVac ($CVAC), and Arcturus Therapeutics ($ARCT). These companies have been investigating mRNA vaccines against other coronaviruses, including MERS-CoV and SARS-CoV.
Source: Corbett et al. 2020
When the novel coronavirus sequences were released, it was a simple matter to modify the mRNA transcript to encode the spike (S) protein for SARS-CoV-2 (the pathogen causing COVID-19) (ref). Moderna was able to launch a phase 1 trial just 66 days after the release of the sequences. The authors employed double proline mutations at the N-terminal cap of the central helix, which have been found to stabilize the spike protein of other coronaviruses (ref). These mutations appear to keep the S protein in a prefusion conformation and enhance its expression levels and immunogenicity in animals. Given the high degree of homology, and that helices have a preference for prolines at the N-terminus (ref), this S protein configuration could be a generalizable approach to creating robust coronavirus-specific vaccines.
Source: Corbett et al. 2020
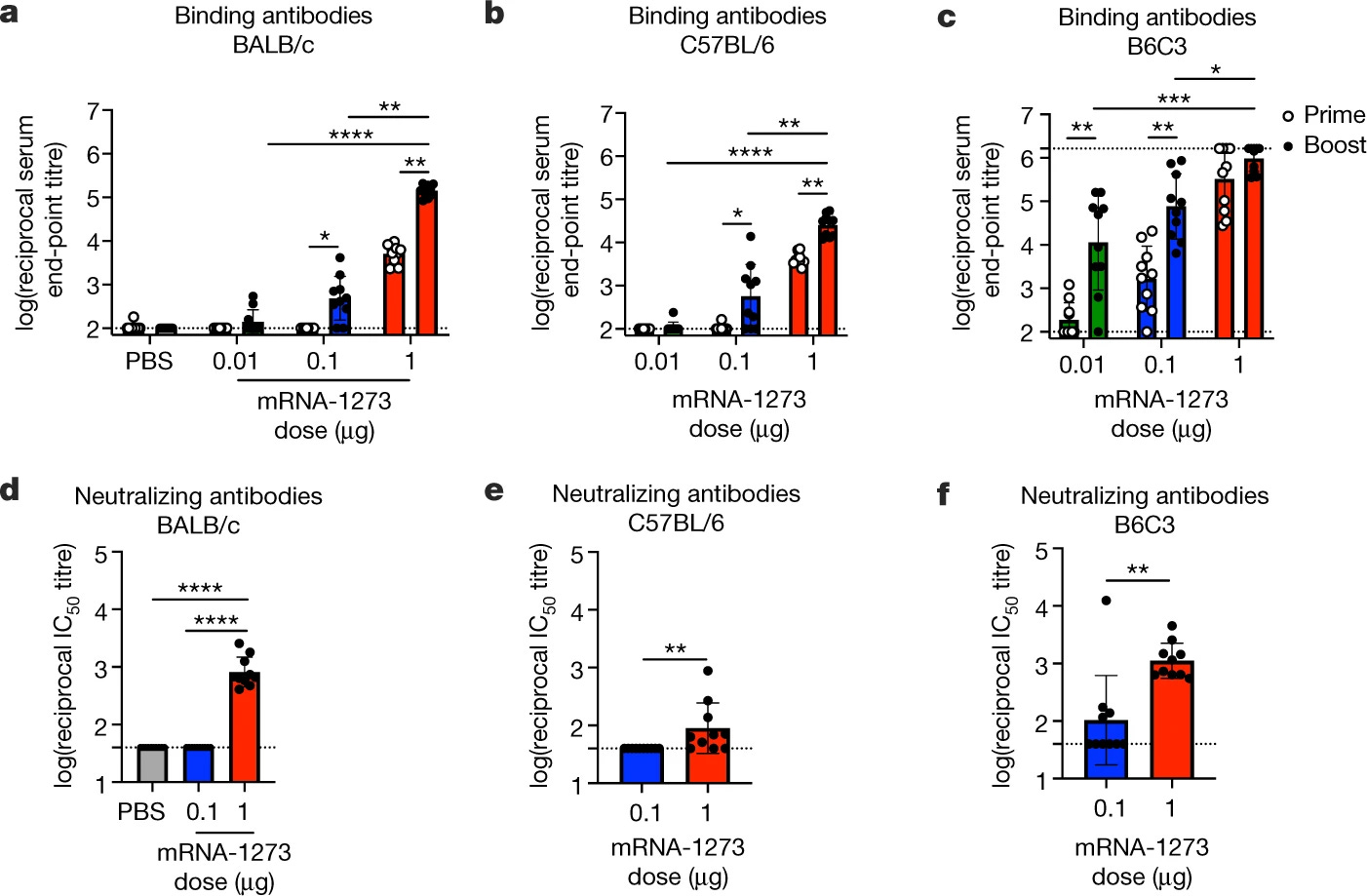
In the animal studies, mRNA-1273 was found to elicit binding and neutralizing antibodies in a dose-dependent manner against pseudoviruses (lentiviruses expressing the S protein of SARS-CoV-2, therefore BL3 containment is not necessary). The authors also demonstrated that mRNA-1273 protected against pseudoviruses expressing the S protein with D614G variation, which was determined to enhance the infectivity of SARS-CoV-2 and is rapidly spreading across the world (ref). The authors also considered the potential for vaccine-associated enhanced respiratory disease (VAERD), which has been associated with TH2-biased immune response. The distribution of helper T cells between TH1 and TH2 were evaluated on the basis of immunoglobulins specific for the S protein. IgG2a and IgG2c levels are surrogates for TH1 response, and IgG1 level is a surrogate for TH2 response. The authors state that TH1:TH2 responses were balanced, but a close look at the supplementary data seemed to indicate a bias towards TH2 response. Interestingly, the phase 1 trial results showed that, through cytokine assays, mRNA-1273 induced TH1-biased response in humans (ref). However, It is not clear whether T cell response in mice is necessarily indicative of T cell response in humans.
Source: Jackson et al. 2020
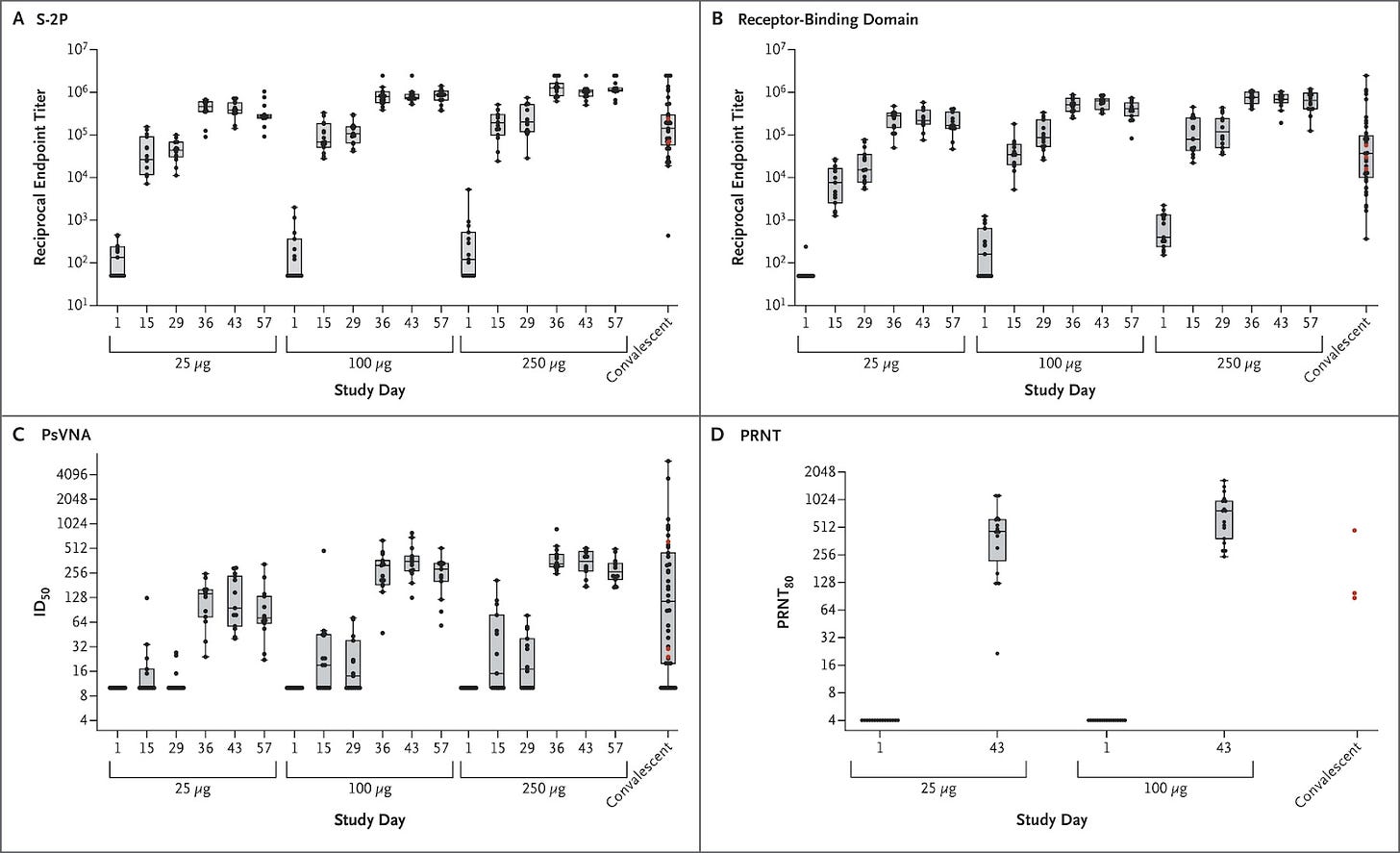
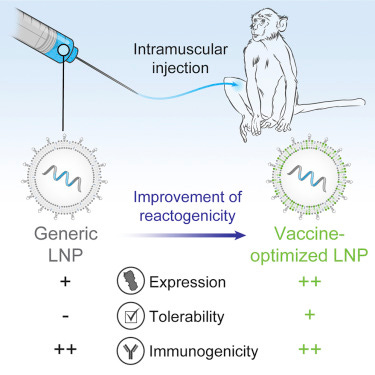
More generally, the phase 1 trial demonstrated that mRNA-1273 was safe and well tolerated, and that it elicited dose-dependent antibody responses. As expected, there was more reactogenicity following the second dose. In the highest dose (250 μg) cohort, three participants (21%) had severe adverse events. STAT News reported an interview with a trial participant, Ian Haydon, who experienced a high fever after receiving the second dose (ref). Fortunately, he recovered within a day.
Source: Hassett et al. 2019
In addition to optimizing the mRNA transcript, significant work has been done to engineer the lipid nanoparticles (ref). Compared to DNA vaccines, which require delivery to the nucleus via electroporation, mRNA vaccines can be delivered to the cytosol via lipid nanoparticles. The authors tested various ionizable lipids while maintaining the same lipid-nitrogen-to-phosphate ratio and molar composition of components. The authors then encapsulated mRNA transcripts for firefly luciferase and the H10N8 influenza hemagglutinin (HA) antigen to determine protein expression and immunogenicity within the same animal. Interestingly, there was a weak, if any, correlation between protein expression and immunogenicity.
Source: Evaluate Vantage report
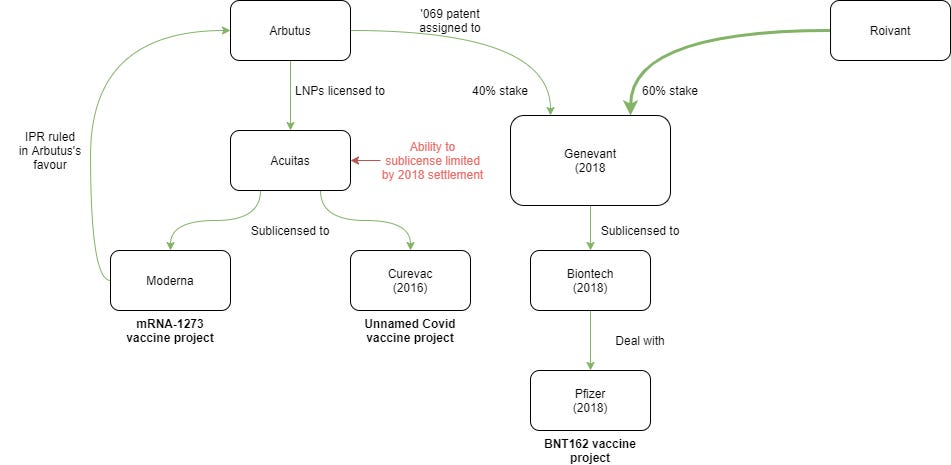
Intriguing, common lipid nanoparticle technology may be shared among the major mRNA vaccine companies (ref). In July, Moderna lost a challenge against Arbutus Biopharma for its US patent 8,058,069 (i.e. ‘069) (ref). Moderna subsequently claimed that these proceedings commenced before the development of their COVID-19 vaccine, and that the lipid nanoparticle formula used in mRNA-1273 did not rely on the Arbutus patents (ref). Regardless, the competing mRNA vaccine companies are quickly capturing neighboring IP space on lipid nanoparticle formulations.
Pandemic economy and the accelerated study of COVID-19 vaccines
Historically, periods of warfare greatly tilt the economy towards extensive defense production and advances in technology; this is known as war economy (ref). This year, the COVID-19 pandemic has ignited international mobilization for effective vaccines and anti-infective treatments. This pandemic economy has many similarities to a war economy. During times of conflict, the government tends to elevate spending tax dollars on defense and competition in the private sector intensifies. Likewise, Operation Warp Speed was launched in May with the objective of accelerating pharmacologic interventions (e.g. vaccines, therapeutics, diagnostics) development in the private sector using taxpayer funds (ref). The competition in the pharmaceutical industry also created significant pressure to make better products at an affordable cost. Notably, vaccine pricing has been a major focus of media attention (ref).
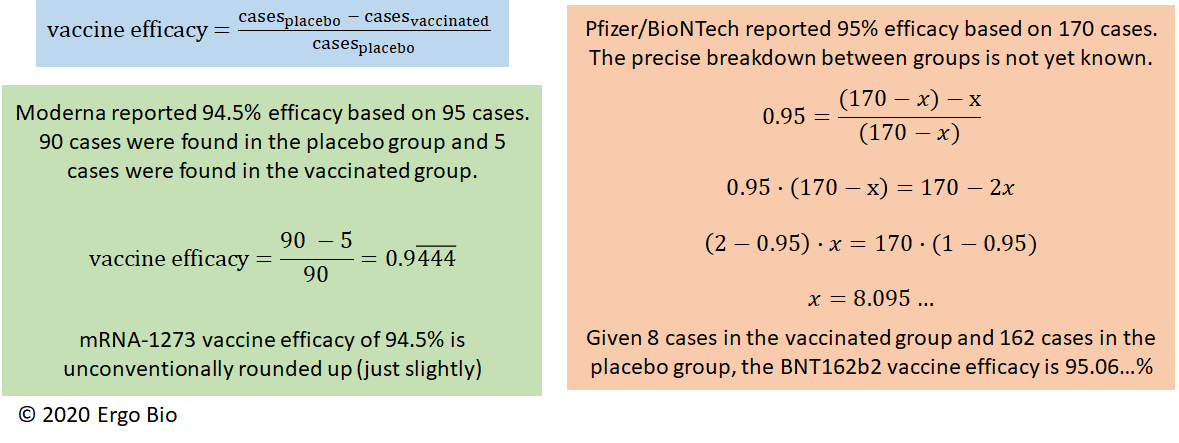
The efforts driven by the pandemic economy have yielded outstanding results. Including the rapid repurposing of remdesivir by Gilead Sciences (see related post) and other therapies, there have been numerous vaccine candidates under clinical investigation. Three companies have posted positive interim analyses for vaccine candidates in phase 3 trials, two of which are mRNA based. The readouts are based on the proportionate reduction in confirmed cases of COVID-19 in vaccinated patients to the confirmed cases of COVID-19 in patients receiving placebo. Moderna reported that two full doses of mRNA-1273 offered a vaccine efficacy of 94.5% (ref). Specifically, they reported that 95 cases were observed with 90 from the placebo group and 5 from the vaccinated group. Back-of-the-envelope calculations indicate that Moderna unconventionally rounded up to 94.5%. Pfizer and BioNTech reported that two full doses of BNT162b2 offered a vaccine efficacy of 95%. This was based on 170 cases, but the exact distribution was not described. Back-of-the envelope calculations indicate that there were 162 cases from the placebo group and 8 cases from the vaccinated group.
AstraZeneca also reported positive results for AZD1222 (chimp adenovirus) based on 131 cases, but two dosing regimens were investigated (ref). Two full doses offered a vaccine efficacy of 62%, whereas a half dose followed by a full dose offered a vaccine efficacy of 90%. Mainstream media has reported the average efficacy of 70% (which is also the fault of AstraZeneca’s press release), but aggregate value is not appropriate given the distinct dosing regimens. Surprisingly, the second dosing regimen was actually a mistake and there was no intention to test it (ref). At present, it is not known why a smaller dose yielded greater efficacy. It could be related to geographical distribution, as each country and even state/province have experienced disparate levels of COVID-19 cases. Generally, there has been substantial criticisms regarding the manner in which AstraZeneca conducted its clinical trials.

What lies ahead for COVID-19 vaccines
Although limited, the efficacy data reported by Moderna, BioNTech (and Pfizer), and AstraZeneca appears promising and it is likely that these vaccines will receive emergency use authorization (and eventually regulatory approval). It is important to note that with additional data (i.e. more cases from either placebo or vaccinated group), the vaccine efficacy values could change dramatically. Complicating the evaluation are geographical variations for the adoption of social distancing, and the ever-changing policies enacted at the county and state levels. Another factor could be that typical trial participants exhibit distinctly different social behaviors than the broader population.
It is also important to note that there is evidence for pre-existing immunity against COVID-19, specifically that SARS-CoV-2 cross-reactive T cells have been found in healthy donors (ref). This is not too surprising given that many colds are coronavirus strains. There are concerns that prior exposure to other coronaviruses could facilitate antibody dependent enhancement (ADE) of SARS-CoV-2 infection (ref). Either way, more comprehensive studies on pre-existing immunity are needed, as well as follow-up studies on why some vaccinated participants still became sick with COVID-19.
To resolve the COVID-19 pandemic and effectively deploy the vaccines, distribution and supply chain management will be critical. Regardless of which product has superior efficacy, the massive world-wide demand ensures that all (if safe and effective) will be able to take some market share. At present, the mRNA vaccines appear the most promising and most straight-forward to manufacture and deploy. However, it will be important to keep a close eye on forthcoming peer-reviewed publications and not to rely just on corporate press releases for understanding the safety and efficacy.
Author information
Ergo Bio closely follows innovation in the biotechnology space and evaluates interesting drugs and deals. It is run by Vandon T Duong (LinkedIn), feel free to connect! I am a biotech enthusiast and a molecular engineer by training. I am also an avid consumer of news and research around precision medicine.
Ergo Bio pages
Disclaimer
This article serves informational purposes only and should be treated strictly as educational material, not as investment recommendation or legal advice. The information presented may be inaccurate or out-of-date. The contributing authors and editors disclaim liability for any errors or omissions. Any opinions expressed may change without notice. Ergo Bio LLC reserves all rights to the content generated through this resource hub (Ergo Bio Insights).